Beginner's Guide to Rockets
Charles and Gay- Lussac's Law
Answers
Answers
will vary. Suggested answers are shown below:
1. Which variable
is plotted on the graph's vertical axis? temperature
2. Which variable
is plotted on the graph's horizontal axis? volume
3. What is
the pressure in kilopascals? 1.5
4. The red
plunger is used to exert pressure on the gas molecules in which colored
area? yellow
5. Complete
the table below as you watch the animated gas lab:
TEMPERATURE
|
VOLUME
|
|
250
|
2.5
|
|
350
|
3.5
|
|
450
|
4.5
|
6. What do
you predict the temperature will be when the volume is 5.5? 550
K
7. Sketch the
completed temperature-volume graph: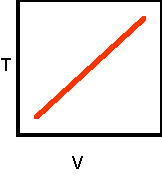
8. Describe
what is used to increase the temperature. A
Bunsen burner is used to heat the gas in the container.
9. Sketch the
completed volume-temperature: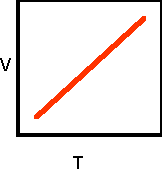
10. Write
the formula equation for Charles and Gay-Lussac's Law. V
= CT
11. Write the
equation for Charles and Gay-Lussac's Law in words. For
a given mass at constant pressure, volume is directly proportional to
temperature.
12. In the
animated gas lab, the unit of temperature is Kelvin.
13. What are
the units of volume used in this lab? cubic
meters
14. Predict
what the volume in this lab would be if the temperature were 525 K. 5.25
15. Predict
what the volume in this lab would be if the temperature were 275 K. 2.75
16. State Charles
and Gay-Lussac's Law in your own words: When
a fixed amount of gas is heated in a closed container, as the temperature
increases the volume also increases.
17. Describe
what happens to the balloons. The volume of
the balloon on the bottle in the hot water bath increases as the temperature
of the air in the balloon increases. The volume of the unheated air in
the other balloon stays the same.
18. How does
your experience with the balloons compare to the animated gas lab? Both
show that as temperature increases, volume of gas increases.
Related Sites:
Teaching Standards
Activity
Worksheet
Rocket Index
Rocket Home
Exploration Systems Mission Directorate Home
|
