Inflating Balloon
Material List:
- a balloon
- a narrow necked soda bottle
- a pan of hot water
- a pan of ice water
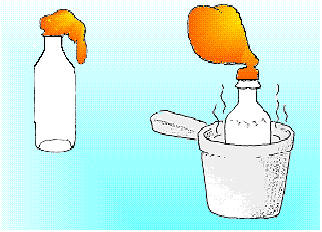
Procedure:
Take the balloon and stretch it gently over the lip of the soda
bottle. Place the bottle into the pan of hot water and let it stand
for a few minutes. To reverse the outcome, place the bottle into the
pan of ice water and let it stand for a few minutes.
Scientific Principle:
This activity demonstrates that the air inside the soda bottle
expands when heated causing the air molecules to move faster and get
farther apart. This is what causes the balloon to inflate. Warm air
is less dense than cold. Since warm air takes up more space than the
same amount of cold air it makes the balloon inflate.
