Thermodynamics is a branch of physics
which deals with the energy and work of a system.
Thermodynamics deals with the
large scale response
of a system which we can observe
and measure in experiments.
As aerodynamicists, we are most interested in the thermodynamics of
propulsion systems
and
high speed flows.
The propulsion system of an aircraft generates
thrust
by accelerating a working fluid, usually a heated
gas.
In response to Newton's
third law
of motion, the propulsion system and the aircraft
are accelerated in the opposite direction.
To understand how a propulsion system works, we must
study the basic thermodynamics of the working fluid.
The working fluid of a propulsion system is a gas.
Gases have various
properties that we can observe with our
senses, including the gas
pressure p,
temperature T,
mass, and
volume V
that contains the gas.
Careful, scientific observation has determined that these
variables
are related to one another, and the values of these
properties determine the
state
of the gas. The
first and
second
laws of thermodynamics define two additional variables,
enthalpy and
entropy S,
which can also be used to describe the state of a gas.
A thermodynamic process, such as
heating or
compressing the gas,
changes the values of the state variables in a
prescribed manner. The total
work and heat transferred to a gas
depend on the beginning and ending states of the gas and
on the process used to change the state.
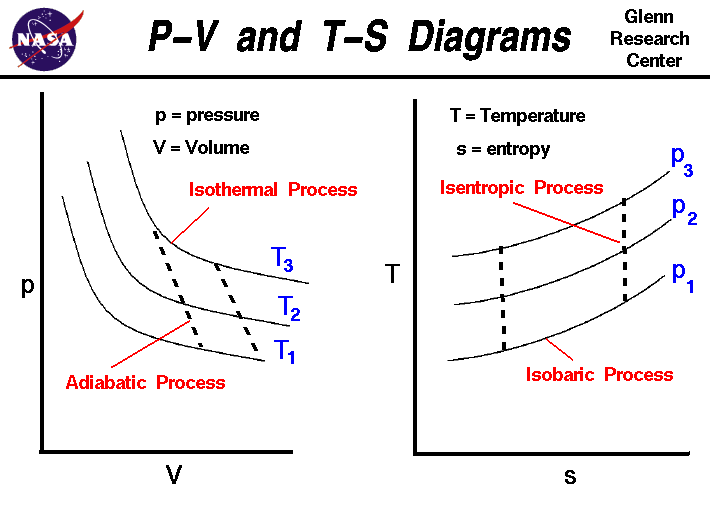
It is useful to plot the changes in the state of a gas during a
thermodynamic process. On the figure we show two types of plots
that are used to describe changes of state. On the left we have
plotted the pressure versus the volume, which is called a p-V diagram.
On a p-V diagram, lines of constant temperature curve from the
upper left to the lower right. A process performed at constant temperature
is called an isothermal process. During an
adiabatic process no heat is transferred to the
gas, but the temperature, pressure, and volume of the gas change as
shown by the dashed line. As described on the
work slide, the area under a process curve on
a p-V diagram is equal to the work performed by a gas during the process.
On the right of the figure we have plotted the temperature versus the
entropy of the gas. This plot is called a T-s diagram. Lines of
constant pressure curve from the lower left to upper right on a T-s diagram.
A constant pressure process is called an isobaric process and
this type of process occurs in the
combustor
of a
gas turbine engine. During an
isentropic process there is no change in
the
entropy
of the system and the process is reversible. An isentropic process
appears as a vertical line on a T-s diagram. The area under a process
curve on a T-s diagram is related to the amount of heat transferred to
the gas.
It is possible to perform a series of processes, in which the state
is changed during each process, but the gas eventually
returns to its original state. Such a series of processes is
called a cycle and forms the basis for understanding
engines. The
Carnot Cycle
describes the operation of refrigerators, the
Otto Cycle
describes the operation of
internal combustion engines, and the
Brayton Cycle
describes the operation of
gas turbine engines.
P-V and T-s diagrams are often used to
visualize the processes in a thermodynamic cycle
and help us better understand the thermodynamics
of engines.
Activities:
Guided Tours
Navigation..

- Beginner's Guide Home Page
|
