To move an airplane through the air,
we have to use some kind of propulsion system
to generate thrust. The most widely used
form of propulsion system for modern aircraft is the
gas turbine
engine. Turbine engines come in a
variety
of forms, including
turbojets,
turbofans, and
turboprops,
but all of these types of engines have some things in common.
All turbine engines have the
core
components of a
compressor,
combustion section,
and
power turbine which drives the
compressor. The
thermodynamics
of all turbine engines are similar.
To understand how a propulsion system works, we must
study the basic thermodynamics of
gases.
Gases have various
properties that we can observe with our
senses, including the gas
pressure p,
temperature T,
mass, and
volume V
that contains the gas.
Careful, scientific observation has determined that these
variables
are related to one another, and the values of these
properties determine the
state
of the gas.
A thermodynamic process, such as
heating or
compressing the gas,
changes the values of the state variables in a
manner which is described by the
laws of thermodynamics. The
work done by a gas
and the
heat transferred to a gas
depend on the beginning and ending states of the gas and
on the process used to change the state.
It is possible to perform a series of processes, in which the state
is changed during each process, but the gas eventually
returns to its original state. Such a series of processes is
called a
cycle
and forms the basis for understanding engine operation.
On this page we discuss
the Brayton Thermodynamic Cycle which is used in
all gas turbine engines.
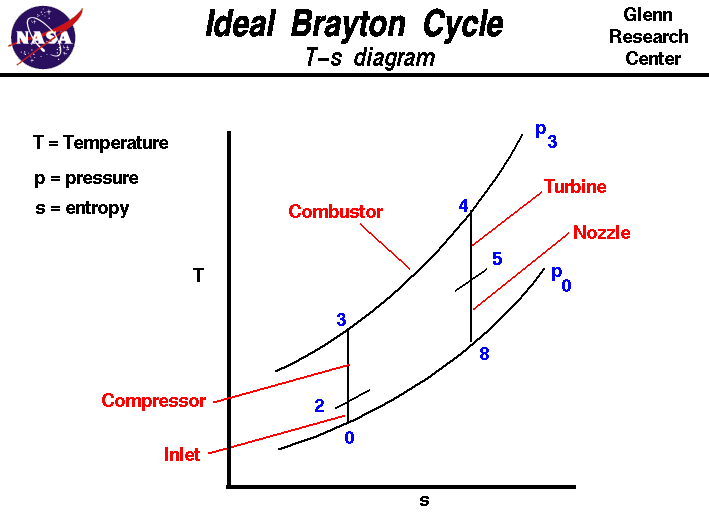
The figure shows a
T-s diagram
of the Brayton cycle. Using the turbine engine
station numbering system,
we begin with
free stream conditions at station 0.
In cruising flight, the
inlet slows the air stream as it is brought
to the
compressor face at station 2.
As the flow slows, some of the energy associated with the
aircraft velocity
increases
the static pressure
of the air and the flow is compressed. Ideally, the compression is
isentropic and the static
temperature is also increased as shown on the
plot. The compressor does
work on the gas and increases the pressure and
temperature isentropically to station 3 the compressor exit.
Since the compression is ideally isentropic, a vertical line on the
T-s diagram describes the process. In reality, the compression is not
isentropic and the compression process line leans to the right because
of the increase in
entropy of the flow. The
combustion process in the
burner occurs at constant pressure from
station 3 to station 4.
The temperature increase depends on the type of fuel used and the
fuel-air ratio.
The hot exhaust is then passed through the
power turbine in which
work
is done by the flow from station 4 to station 5.
Because the turbine and compressor are on the same shaft, the
work done on the turbine is
exactly equal
to the work done by the compressor and, ideally, the temperature change
is the same. The
nozzle then
brings
the flow isentropically back to free stream pressure from
station 5 to station 8. Externally, the flow conditions
return to free stream conditions, which completes the cycle.
The area under the T-s diagram is proportional to the useful work and
thrust
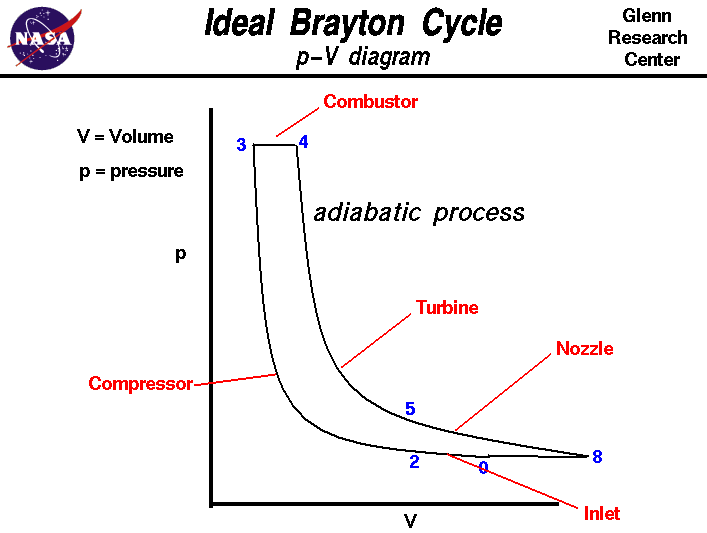
generated by the engine. The p-V diagram for the ideal Brayton Cycle is shown
here:
Activities:
Guided Tours
Navigation..

- Beginner's Guide Home Page
|
