Hot
Air Balloon
SUBJECT: Aeronautics
TOPIC: Lift
DESCRIPTION: An indoor hot air balloon made out of a plastic film dry
cleaner bag. CONTRIBUTED BY: Gregory Vogt (OSU)
EDITED BY: Roger Storm, NASA Glenn Research Center
MATERIALS:
Dry cleaner plastic film bags (select a bag with the thinnest possible plastic
and have several on hand. You may have to experiment with bags of different
thicknesses.)
Several small paper clips
Cellophane tape
Heat source (Blow dryer, sterno, backpacker camp stove, etc.)
Matches
Three feet of aluminum heat duct (if using open flame heat source)
Electric drill (to put holes in the heat duct)
PROCEDURE:
1. Seal any openings and tears in the upper end of the bag with a minimum
of cellophane tape.
2. Attach several paper clips to the plastic around the lower opening. The
number of paper clips to attach is determined by experimentation.
3. If using sterno or some other open flame heat source, prepare the heat
duct by drilling several holes around the base to allow air to flow in.
4. Turn on the blow dryer (or light the Sterno or stove and then set the heat
duct over it) Spread the bag opening wide to capture the rising hot air while
supporting the upper end with your hand. It is best to have assistance in
keeping the bag open so that it does not melt.
5. When the bag is inflated with hot air, test its buoyancy by letting it
go for a moment. If it rises quickly, stand back and let it fly otherwise
continue heating it for a little while longer.
6. If the bag tips over and spills its hot air before it reaches the ceiling,
add a few more paper clips to weigh down the bottom slightly. If the bag will
not rise at all, remove a few clips.
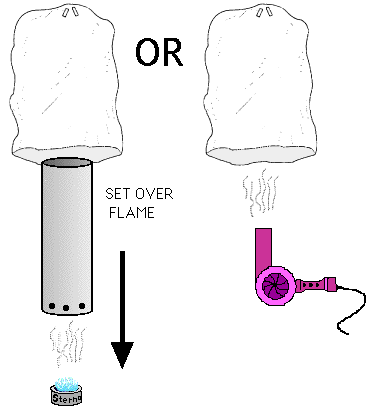
CAUTION: Be careful not to brush clothes or fingers into the flames
or touch the metal heat duct. Keep a fire extinguisher handy if you use flames.
If the bag starts to crumple and melt from the heat, set the blow dryer on
a lower setting or hold the bag farther from the heat source.
DISCUSSION:
Hot air is less dense than cold air. Heat accelerates the motion of the air
molecules causing fewer molecules to occupy the same space as a much greater
number of molecules do at a lower temperature. With fewer molecules, the hot
air has less mass, and therefore is buoyant than an equal volume of colder
air.
Placing the dry cleaner
bag over the heat source captures the hot air and forces out the cooler air
in the bag. The bag becomes a mass of low-density air which floats up-
ward in the higher denser air surrounding it. The paper clips are placed at
the bottom of the bag to keep the open end downward in flight to prevent it
from prematurely spilling the hot air and terminating the flight.
Return to Aeronautics Activities
Return to Aerospace Activities Page
Air Density
